Understand the Label
The communication of safety information is a key responsibility for producers. Like for all end-users served by the soaps, detergents and maintenance products industry, professional customers and operators are entitled to accurate, up-to-date, safe information in order to allow them understand the precautions required in using, handling and storing products.
The product label contains basic information on how the product should be safely handled, used and disposed of. Safety information on labelling is now regulated across Europe, but the market preference for concentrated products has had a dual effect. It reduces the size of the available label space and sometimes gives rise to a higher safety classification requiring more detailed hazard information. Information on the regulations can be found on the website for the European Chemical Agency.
i) CLP Hazard Pictograms
Cleaning products are regulated under the Classification, Labelling and Packaging (CLP) Regulations and the following hazard pictograms are a white diamond with a red border and also need to carry ‘signal words’ such as Danger or Warning and hazard statements such as irritating to eyes or harmful if swallowed. The information required must be in the language of the country the product is sold in.
 IRRITANT/HARMFUL
IRRITANT/HARMFUL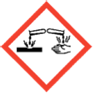 CORROSIVE
CORROSIVE HIGHLY/EXTREMELY FLAMMABLE
HIGHLY/EXTREMELY FLAMMABLE OXIDISING
OXIDISING HAZARDOUS TO THE ENVIRONMENT
HAZARDOUS TO THE ENVIRONMENT
ii) Directions for use
The manufacturers must also provide sufficient information on how to use the product to ensure the product performs properly.
iii) Use Pictograms
Voluntary use pictograms have significantly helped to overcome the multilingual communication diversities across Europe and also reduced the training time needed to use a product. In particular, the European Cleaning Products Industry (AISE – www.aise.eu) has developed two sets of pictograms for use on product labelling and in training/information documents and some examples are shown below. Companies are free to develop their own icons for their products.
The first AISE set of pictograms promotes the correct and best use of products, indicating the actual application area and the application process.
Examples:



The second set indicates the application area for products used in restaurants and catering. They specifically aim at facilitating the proper identification of many products.




iv) Ingredients
For detergents intended to be used in the industrial sector, and not made available to members of the general public, the ingredients do not have to be listed if the equivalent information is provided by means of technical data sheets, safety data sheets, or in a similar appropriate manner. Certain products also require the statement “Safety data sheet available for professional user on request.”.
v) Barcode
A barcode is normally included in the label.
vi) Helpline and manufacturers details
The manufacturer’s name, address and telephone number must be included on the label.
vii) Inner container/pack / outer pack label requirements
The information required by the CLP Regulations should be shown on each layer of packaging unless it is exempted by using any of the International Transport Regulations.
viii) UKCA marking
The UKCA (UK Conformity Assessed) marking is a UK product marking that is used for goods being placed on the market in Great Britain (England, Wales and Scotland). It covers most goods which previously required the CE marking, known as ‘new approach’ goods.
The UKCA marking came into effect on 1 January 2021. However, to allow businesses time to adjust to the new requirements, you will still be able to use the CE marking until 1 January 2023 in most cases.
The UKCA marking applies to most goods previously subject to the CE marking. It also applies to aerosol products that previously required the ‘reverse epsilon’ marking.
The technical requirements (‘essential requirements’) you must meet – and the conformity assessment processes and standards that can be used to demonstrate conformity – are largely the same as they were for the CE marking.
The UKCA marking alone cannot be used for goods placed on the Northern Ireland market.
More detailed guidance can be found on the gov.uk website https://www.gov.uk/guidance/using-the-ukca-marking
ix) Products Used with Medicinal Devices (UKCA Marked)
When cleaning a medical device only an approved product should be used. This will carry the UKCA mark which is seen as a declaration by the manufacturer that the product meets all the appropriate provisions of the relevant legislation including those relating to safety and where required has been assessed in accordance with these. Further information can be found at the Medicines and Healthcare products Regulatory Agency website.

x) Defra Approved Disinfectants
Defra has in place a statutory mechanism under The Diseases of Animals (Approved Disinfectants) (England) Order 2007. This allows veterinary disinfectants to be placed on an approved list for the control of different diseases if they demonstrate efficacy in defined laboratory testing. The Animal Health and Veterinary Laboratories Agency (AHVLA) delivers the scheme on behalf of Defra.
Veterinary disinfectants are a vital component of animal disease control strategies, both in the everyday prevention of disease and in controlling disease spread during epidemic outbreaks. Only approved disinfectants may be used during epidemic outbreaks. The label for approved products will give the specific dosage/dilution instructions and these should be followed to ensure effective treatment. See the Defra website for more details.
Further Guidance and Examples
AISE has further information about safe use and hazard information.